INPUT
The “Input” module is the starting point for all consecutive steps of the Toolbox workflow.
The first module provides different ways to specify the identity of the target substance as well as the property under consideration.
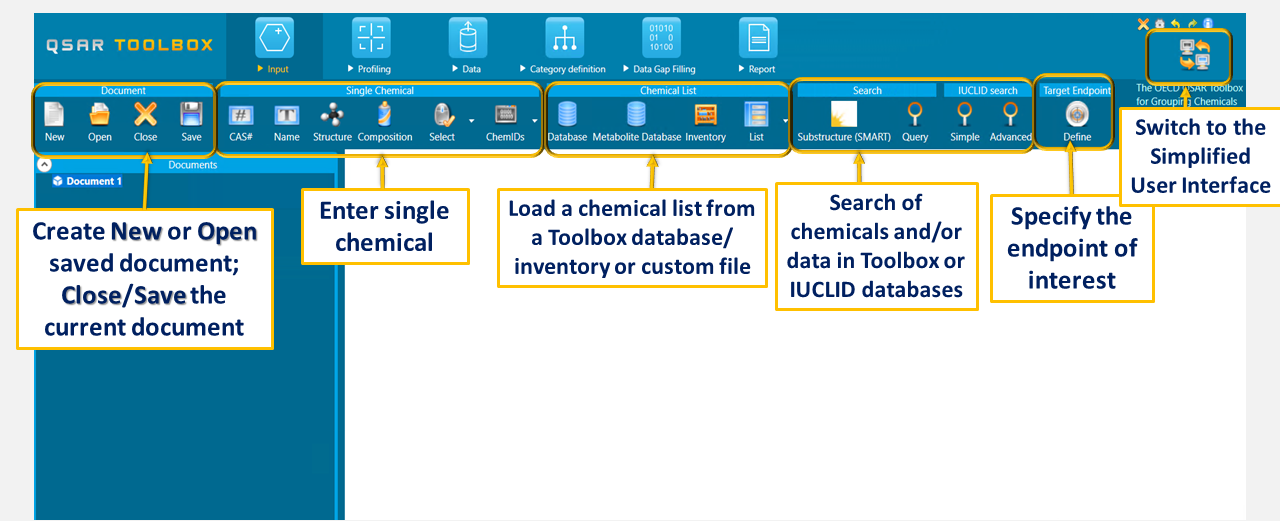
IN THE INPUT THE USERS ARE ABLE TO:
1. Open a new or already saved document
2. Close or save the current document
3. Load a single target chemical – by CAS, Name, Structure – paste SMILES or drawing (including mixtures), select from a file
4. Load a list of chemicals – database, metabolism database, inventory, custom file
5. Customized search – searching chemicals and/or data within the Toolbox databases. One or more than one criteria combined with logical operators (AND, OR, NOT) could be used.
6. Search in IUCLID databases – searching chemicals and/or data within the ECHA and/or other custom IUCLID databases imported in Toolbox. Simple and advanced (specifying more criteria on the composition/data) search could be applied.
7. Define target endpoint – specifying the endpoint of interest, the system will further indicate the suitable databases/profilers/metabolic simulators that could be used.
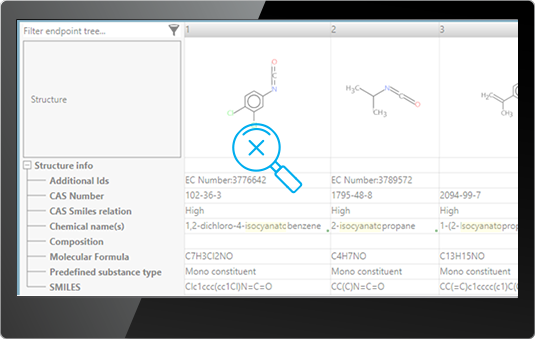
![]() Chemical ID – you could search chemicals based on their CAS RNs or Names
Chemical ID – you could search chemicals based on their CAS RNs or Names
E.g. Searching for chemicals having names containing “isocyanato”
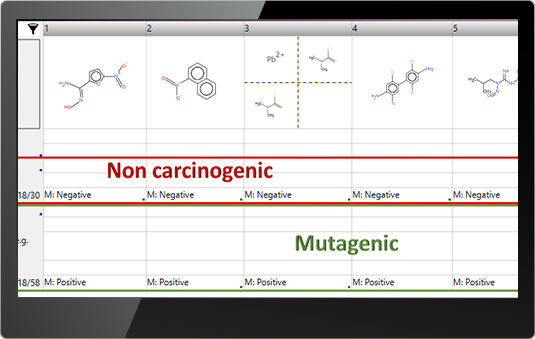
![]() Data – you could search for all available data for an endpoint(s) of interest or for chemicals having specific experimental results
Data – you could search for all available data for an endpoint(s) of interest or for chemicals having specific experimental results
E.g. Searching for chemicals with positive skin sensitization data
E.g. Searching for mutagenic but not carcinogenic chemicals
![]() Physico-chemical properties – you could search for chemicals with a given property (calculated or by experimental data) with defined threshold(s)
Physico-chemical properties – you could search for chemicals with a given property (calculated or by experimental data) with defined threshold(s)
E.g. Searching for not bioavailble and lipophilic (logKow > 4) chemicals
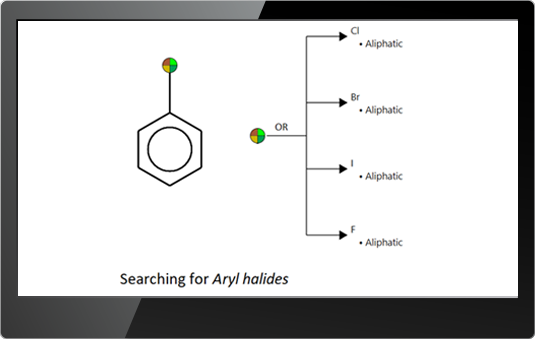
![]() Subfragments – searching for specific part of the molecules using the SMARTS language – The SMARTS (SMiles ARbitrary Target Specification) is an extension of SMILES code designed to describe substructure patterns used for fragment matching www.daylight.com/dayhtml/doc/theory/theory.smarts.html.
Subfragments – searching for specific part of the molecules using the SMARTS language – The SMARTS (SMiles ARbitrary Target Specification) is an extension of SMILES code designed to describe substructure patterns used for fragment matching www.daylight.com/dayhtml/doc/theory/theory.smarts.html.
A special editor is developed to facilitate the users in writing SMARTS.
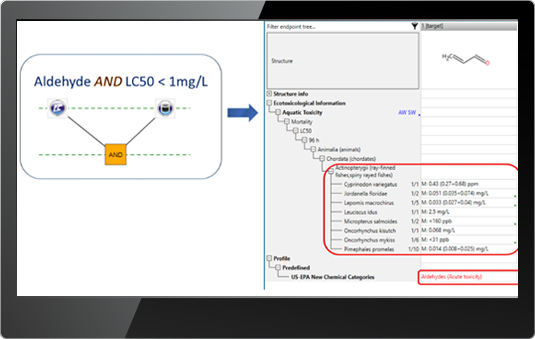
![]() Profiling results – allows to search for chemicals having specific functionality or mechanisms according to a selected profiler.
Profiling results – allows to search for chemicals having specific functionality or mechanisms according to a selected profiler.
E.g. Searching for aldehydes having experimental LC50 < 1mg/L
![]() Structural similarity
Structural similarity
E.g. Searching for structures with similarity above 65% to my target structure
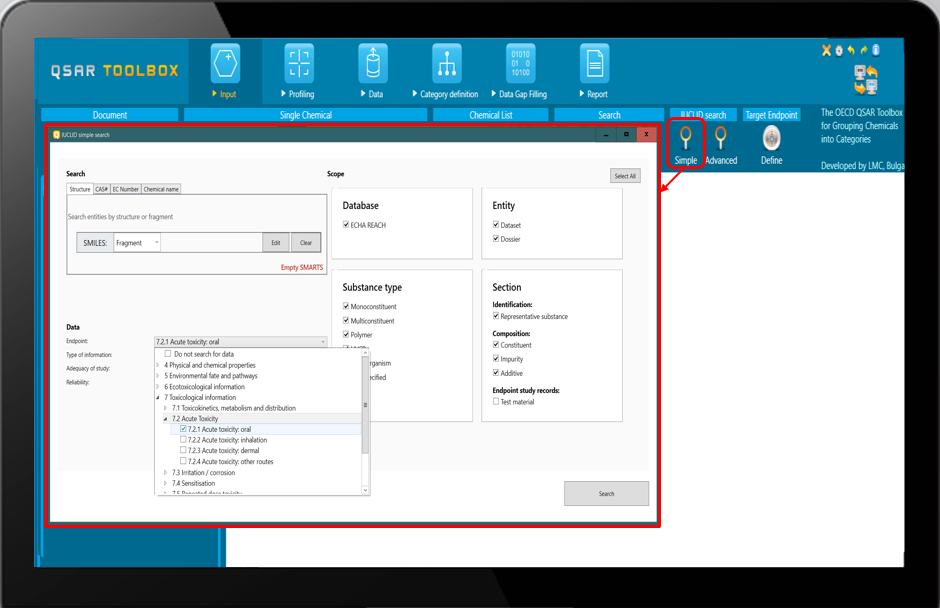
![]() IUCLID search – searching for specific substances having known additives, impurities, etc. and/or available data in ECHA REACH database or in the imported custom IUCLID databases.
IUCLID search – searching for specific substances having known additives, impurities, etc. and/or available data in ECHA REACH database or in the imported custom IUCLID databases.
Use the Simple IUCLID search for more general searches or use the Advanced IUCLID search for more precise queries.
E.g. Searching for substances having impurity with isocyanate fragment